NEWS
Project Twenty21 on track for summer launch
The largest European study of its kind aims to provide evidence of the efficacy of medical cannabis for a range of conditions.
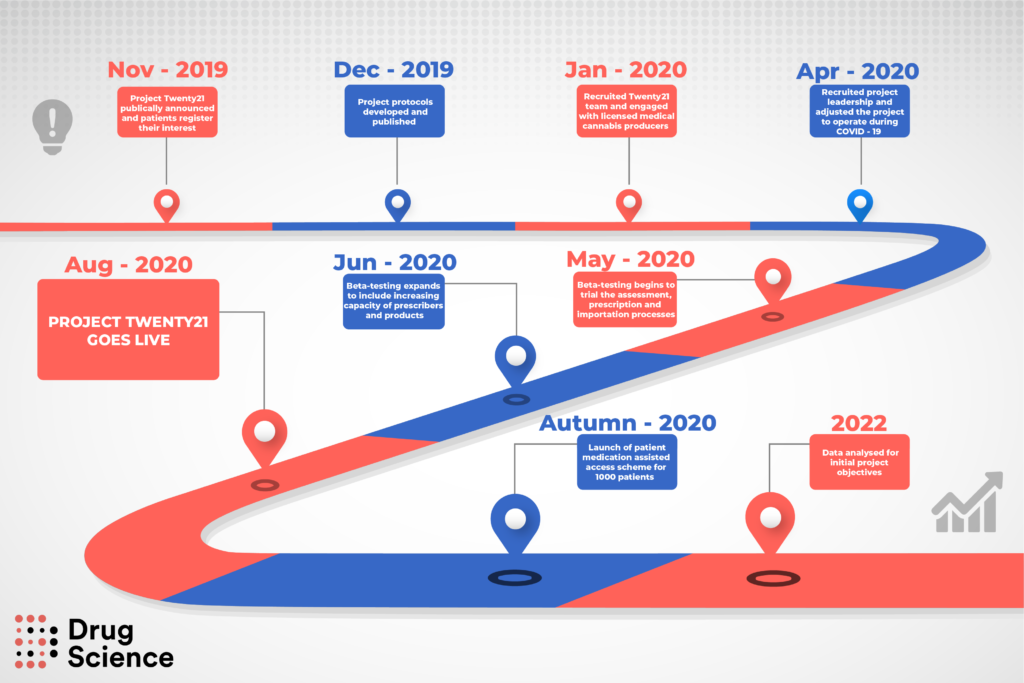
The landmark Project Twenty21 medical cannabis register is still on track to launch to patients this summer.
The largest European project of its kind aims to provide a vast database of evidence gathered from medical cannabis patients with a range of conditions, from anxiety to chronic pain.
The team at charity Drug Science hopes that the data will inform wider prescribing of medical cannabis on the NHS.
The COVID-19 outbreak caused ‘major disruption’ to the development of the project. However, virtual appointments increased patient access and reduced the cost of private consultations.
“The Project Twenty21 team completed the first test phase of the registry at the end of May 2020.
“Now the focus of the project is to increase the number of doctors who can prescribe medical cannabis, aiming for an official launch to patients in summer 2020.”
Project Twenty21 has partnered with five licensed cannabis producers who can provide various medical cannabis formulations at a significantly reduced cost of £150 per month.
There are currently only two types of medical cannabis available on the NHS.
Sativex, a 1:1 CBD:THC spray, can be prescribed for MS-related muscle spasticity where other treatments have been unsuccessful.
Epidyolex is used to treat Lennox-Gastaut syndrome and Dravet syndrome: two rare types of epilepsy.
Meanwhile, nabilone, a synthetic drug that mimics the effects of THC, is prescribed by specialist doctors to chemotherapy patients when they feel sick or vomit.
A Freedom of Information (FoA) request by the Guardian newspaper in June revealed that no new NHS patients had been prescribed full-extract cannabis oil since legalisation in 2018.
Drug Science Scientific Committee chair, Professor David Nutt, said:
“I suspect many doctors are afraid of cannabis after years of fear-mongering and just want to be spoon-fed by the pharmaceutical industry”










